
Turn aesthetic course ideas into high-impact, fully executed labs
Applied Anatomy partners with your organization to set up and coordinate advanced aesthetic training programs with precision and clinical integrity

Your trusted professional full-service lab partner
With over 10 years of experience organizing mobile cadaveric labs, Applied Anatomy has supported industry leaders in aesthetics, professional training boards, and university-affiliated medical groups across five provinces in Canada and various states in America, with continued work throughout North America.
We’d be happy to walk through your specific needs.
Toronto, Ontario serves as our headquarters, providing full-service coverage across North America.
Who we work with
Applied Anatomy supports organizations that need cadaveric or tissue-based labs executed correctly, compliantly, and without disruption, including:
✓ Medical education boards and training institutions
✓ Aesthetic and surgical course providers
✓ Industry groups and medical device companies
✓ Educational filming and instructional course teams
✓ Event organizers hosting advanced clinical labs
*Many of the programs we support involve sensitive logistics, proprietary course structures, or institutional policies, client confidentiality is a priority. For this reason, company names are not publicly listed. Referrals and references are available upon request..
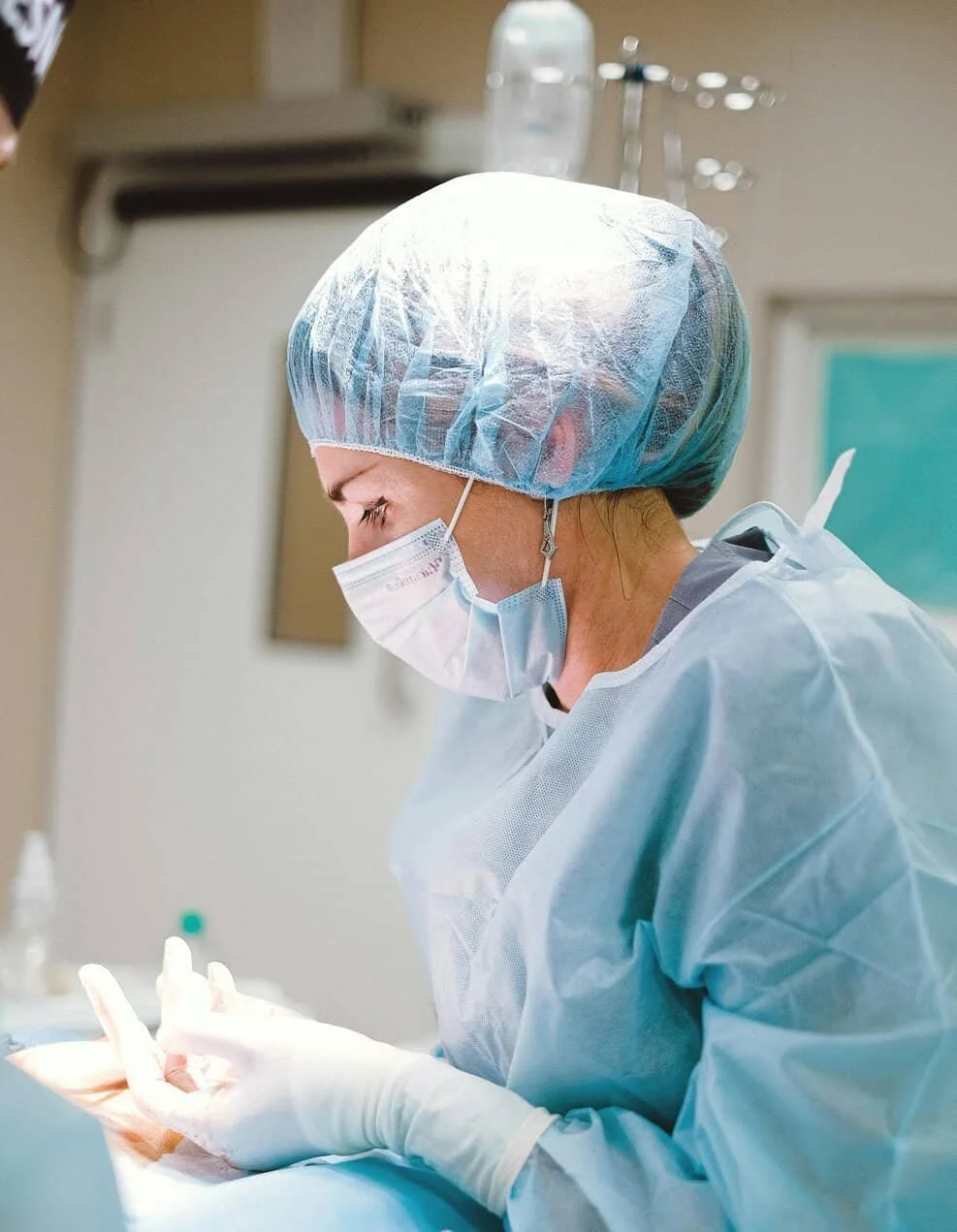
End-to-end lab coordination
1. Initial Consultation & Program Review
We start with a brief consultation to understand your course goals, location, timeline, and facility requirements. This allows us to identify regulatory considerations, tissue needs, and logistical constraints early—before problems arise.
2. Planning, Compliance & Coordination
Applied Anatomy manages the behind-the-scenes complexity, including tissue sourcing, customs clearance (if required), funeral home coordination, PPE planning, instrumentation, and alignment with local, provincial, or state regulations.
3. Lab Setup & On-Site Execution
We build and manage the lab from the ground up—creating workstations with proper instrumentation, disposables, and workflow. Our team supports on-site execution to ensure the lab runs smoothly and professionally.
4. Post-Lab Cleanup & Close-Out
Following the lab, we coordinate tissue handling, transport or cremation as required, and complete a thorough cleanup so the facility is returned exactly as it was found.
Our Services
Running a tissue-based lab is complex—and mistakes are costly.
We help organizations overcome challenges such as:
Sourcing appropriate tissue from licensed tissue banks
Customs clearance and cross-border transport of tissue
Coordinating licensed funeral homes for compliant transport to and from lab locations
Navigating state, provincial, and institutional regulations
Meeting PPE, Instrumentation and biosafety requirements for cadaveric labs
Addressing facility manager concerns around hosting tissue-based labs

Contact Us
Planning a lab or program? Applied Anatomy handles the logistics of tissue-based training so your course runs smoothly, stays compliant, and has no surprises. Early planning or final tweaks—we’ll discuss your needs and see if we’re the right fit.
Frequently Asked Questions
-
Yes. Applied Anatomy works with licensed tissue banks across North America and coordinates all required logistics, including sourcing, transport, and documentation in accordance with applicable regulations.
-
Yes. We have experience coordinating labs across provincial and state borders, incuding customs clearance when required.
-
We work directly with facility managers to address biosafety, PPE requirements, regulatory compliance, and post-lab restoration. Our goal is to ensure facilities are comfortable, informed, and fully protected.
-
Yes. We advise on and implement appropriate PPE, necessary instrumentation, and biosafety protocols specific to cadaveric and tissue-based labs.
-
We coordinate tissue transport or cremation as required and complete a thorough cleanup, returning the space to its original condition.
-
We recommend engaging a minimum of 12 weeks in advance to allow sufficient time for compliance review, tissue sourcing, logistics, and facility coordination—particularly for cross-border or multi-day labs. Earlier engagement is strongly encouraged to help ensure smooth, issue-free execution.
Have additional questions? Schedule a free 20-minute consultation and let’s talk through your program needs.
